Abstract
Genetically engineered CD19-redirected chimeric antigen receptor T (CAR T) cells represent a breakthrough immunotherapy for B cell malignancies. However, response rates are variable and many patients face life-threatening side effects such as cytokine release syndrome (CRS) or immune effector cell-associated neurotoxicity syndrome (ICANS). The impact of the patient's immune landscape as a whole - consisting of CAR T cells and non-CAR T immune cells - on patients' outcome is largely unknown. Furthermore, reliable biomarkers to predict response and toxicity are lacking.
To decode the crosstalk of CAR T cells and their circulating immune environment, we used longitudinal, high-dimensional single-cell spectral cytometry and algorithm-guided computational analysis of 47 peripheral blood samples from 19 lymphoma patients, together with 6 healthy controls. Patient samples were harvested one day before (day -1) up to 104 days after CAR T infusion. The generated immune map, based on 69 parameters and 2078 extracted immune features, was enriched with numerous clinical parameters from the lymphoma patients, including response and toxicity data of a follow-up period ≥ 315 days.
When comparing healthy controls and B cell lymphoma patient samples at baseline (day -1 and day 0, pre-CAR T infusion), the two cohorts demonstrated distinct immune profiles, including a strong lymphodepletion and proportional expansion of the myeloid/ dendritic cell fraction in lymphoma patients.
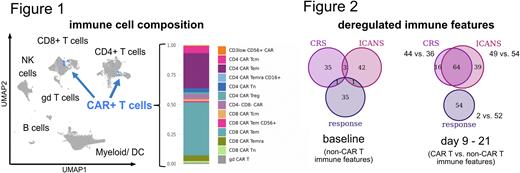
Furthermore, this translational proteomics approach revealed a lymphoma patient-specific, heterogeneous CAR T cell population (Figure 1). However, most of the identified 13 CAR T subsets, such as γδ T cells and CD4 T regulatory cells next to naive/effector/memory T cell populations, appeared in each sample. The temporal composition of the CAR+ T population varied, presenting an inverse relationship over time with a bias of CD8 CAR T cells at early timepoints and dominating CD4 CAR T subsets later on.
When stratifying the overall immune profile at the time of CAR T cell expansion and toxicities (day 9 to 21), immune alterations associated with response to CAR T therapy (best overall response complete remission (CR)) were primarily found in the non-CAR T immune compartments, while CRS- and ICANS-associated signatures affected predominantly the CAR T population (Figure 2; deregulated immune features: p < 0.5 and wilcoxon effect size > 0.6). In particular, higher expression of TCF1 in CD8 CAR T cells and EOMES in CD8 T stem cell memory (Tscm) cells - suggestive for stem-like features that allow expansion/long-term persistence - and CD223 (LAG3) in CD4 CAR T cells marked the immune profile of CR patients. The interplay of CAR T and non-CAR T immune alterations was even more striking in toxicity: both CRS and ICANS were linked to a dysbalance in the CD226/TIGIT-axis with higher expression of the co-stimulatory receptor CD226 together with its regulator EOMES in CD4 and CD8 CAR T cells, whereas the co-inhibitory and competing receptor TIGIT demonstrated reduced levels in several CAR T subsets as well as CD8 T central memory and Tscm cells. In line with the latter result, lower expression of the immune checkpoint CTLA-4 was detected in both CAR positive and negative unconventional T cells, indicating immunopathological signatures in CRS and ICANS are based on imbalanced activating/inhibitory receptor profiles.
Supportive for a pivotal role of the immune environment in CAR T treated patients, stratifying immune signatures associated with patient outcomes were already detected pre-CAR-T-infusion (Figure 2). Towards clinical translation, these altered signatures affecting the T, NK and myeloid/dendritic cell compartment are further investigated and could serve as potential biomarkers predicting patient outcome.
In-depth immune profiling of CAR T treated lymphoma patients allowed us to ultimately define the CAR T specific immune landscape and uncovered a synergy of CAR T and surrounding immune cells in stratifying immune profiles. These results could serve to delineate CAR T-based therapeutic strategies targeting the immunopathologic cascade, aimed to enhance anti-cancer activity and confine toxicity.
.
Disclosures
Alberti:Philogen S.p.A.: Current Employment. Zenz:Roche: Consultancy, Honoraria; Takeda: Consultancy, Honoraria; Gilead: Consultancy, Honoraria; Janssen: Consultancy, Honoraria; Abbvie: Consultancy, Honoraria; Incyte: Consultancy, Honoraria; Janpix: Consultancy, Honoraria. Manz:CDR-Life Inc: Consultancy, Current holder of stock options in a privately-held company; University of Zurich: Patents & Royalties: CD117xCD3 TEA. Müller:Novartis: Consultancy, Honoraria; Kite/Gilead: Consultancy, Honoraria; Celgene/BMS: Consultancy, Honoraria; Janssen: Consultancy, Honoraria. Becher:Numab: Membership on an entity's Board of Directors or advisory committees.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal